
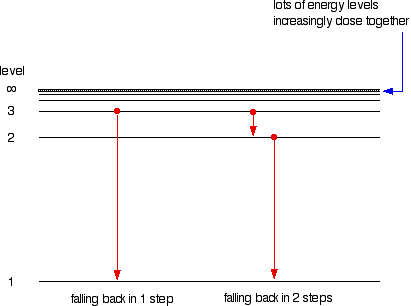
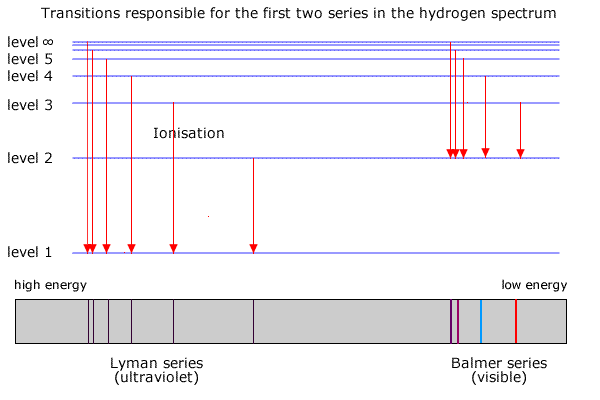
6. The line emission spectrum of hydrogen provides evidence for the existence of electrons in discrete energy levels, which converge at higher energies. – The Atomic Project (SL Chemistry)
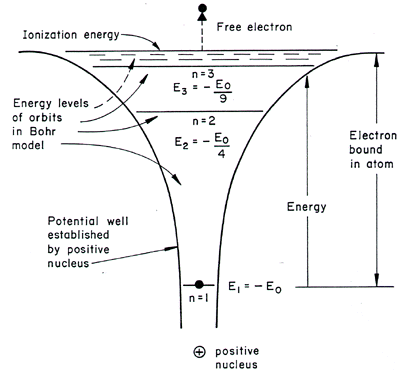
Why are the convergence lines in the UV spectrum used to calculate the ionization energy of hydrogen rather than the convergence line in the visible spectrum? | Socratic
HOW does the line emission spectrum of hydrogen provide evidence for the existence of electrons in discrete energy levels, which converge at higher energies? - Quora
HOW does the line emission spectrum of hydrogen provide evidence for the existence of electrons in discrete energy levels, which converge at higher energies? - Quora
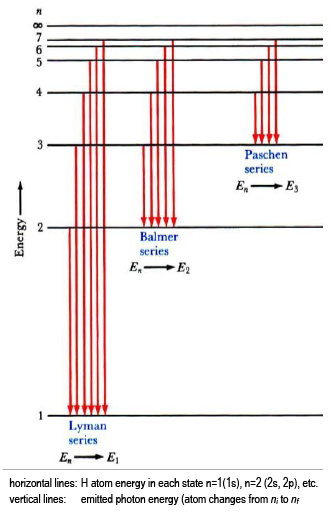
Why are the convergence lines in the UV spectrum used to calculate the ionization energy of hydrogen rather than the convergence line in the visible spectrum? | Socratic

Hydrogen emission spectrum spectroscopy successive ionisation energy patterns related to sub-shells and group of periodic table GCE A Level revision notes


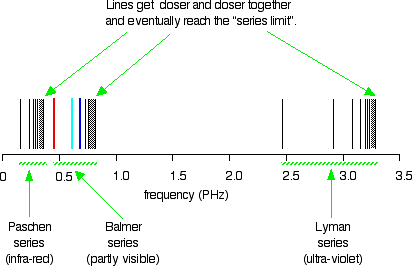
![2.2 The Line Spectrum of Hydrogen [SL IB Chemistry] - YouTube 2.2 The Line Spectrum of Hydrogen [SL IB Chemistry] - YouTube](https://i.ytimg.com/vi/6rHerkru60E/maxresdefault.jpg)
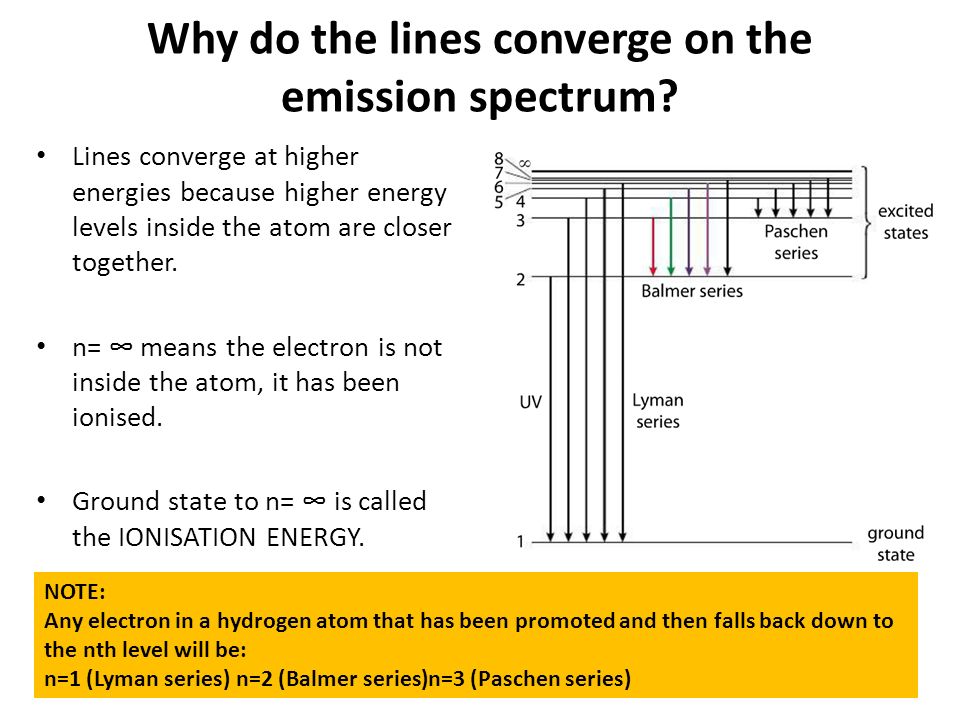


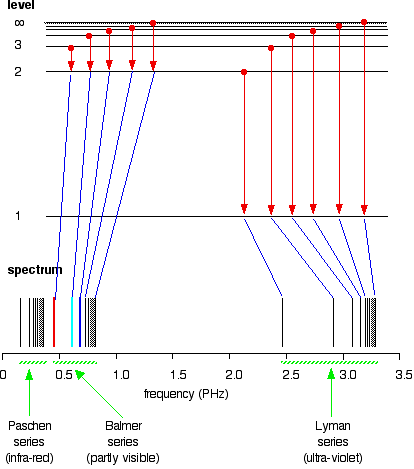

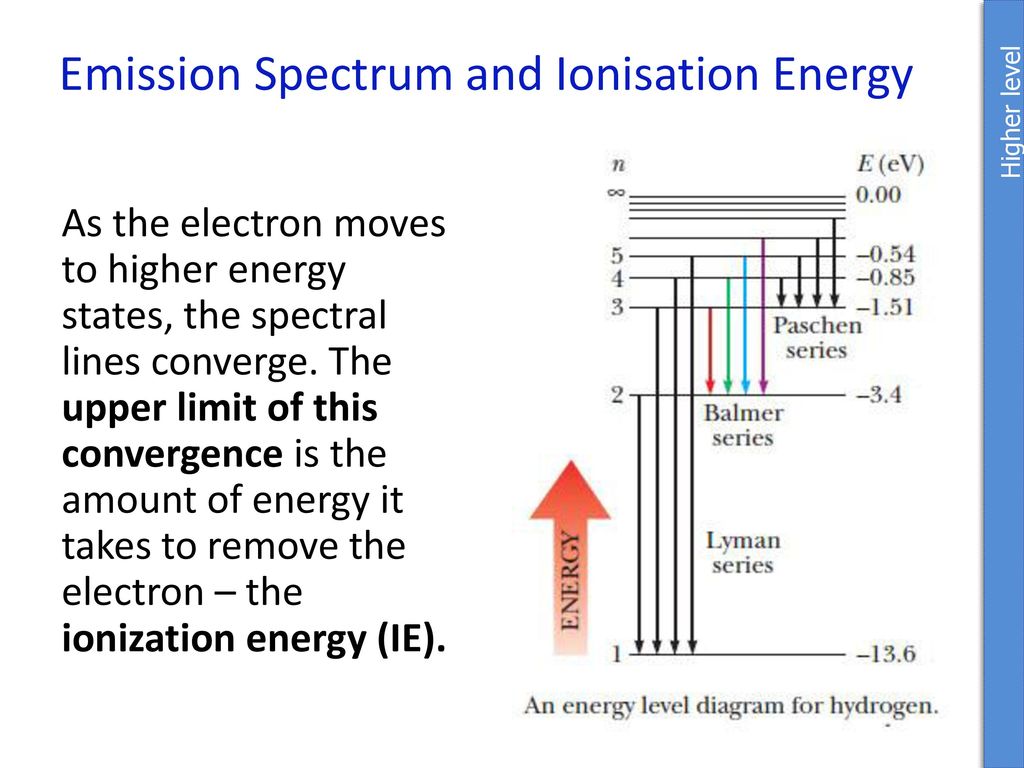

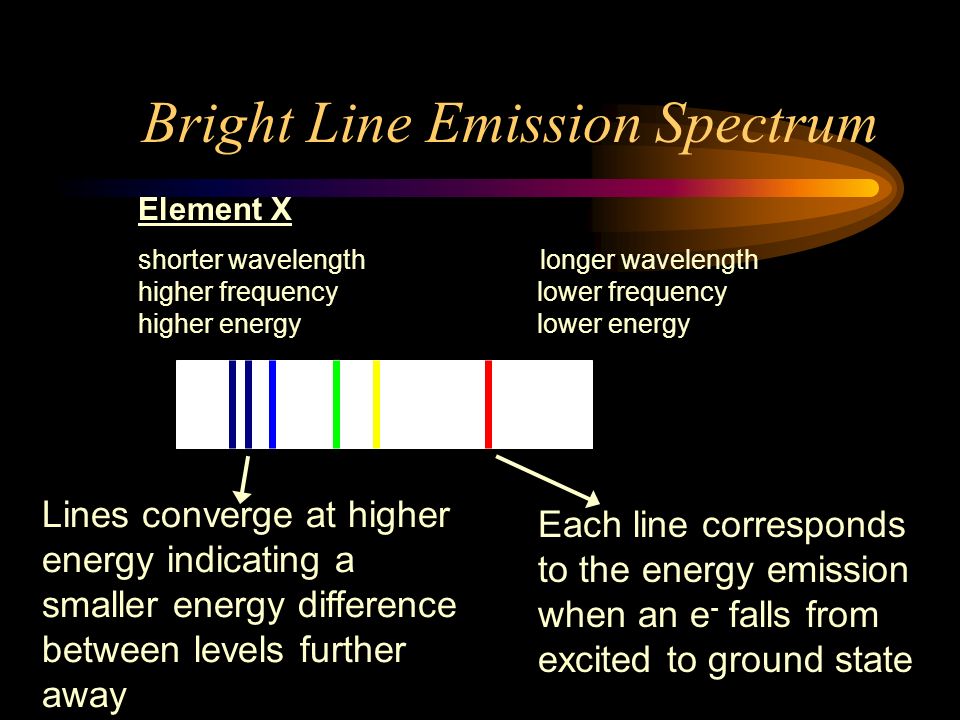


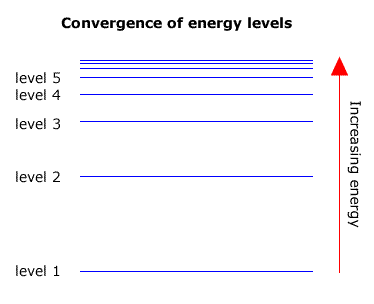
![12.1 Limit of Convergence and Calculations [HL IB Chemistry] - YouTube 12.1 Limit of Convergence and Calculations [HL IB Chemistry] - YouTube](https://i.ytimg.com/vi/rQELfNFsvaQ/maxresdefault.jpg)